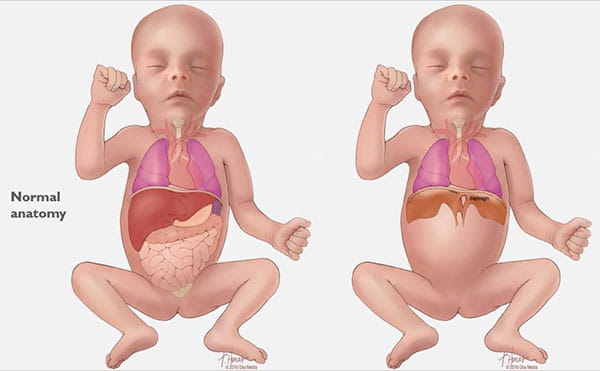
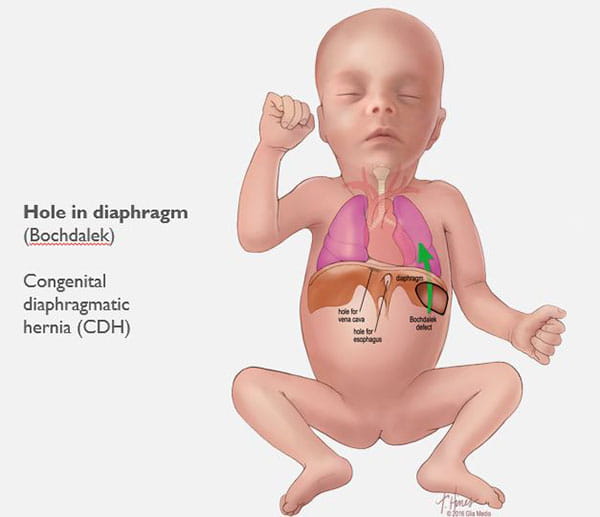
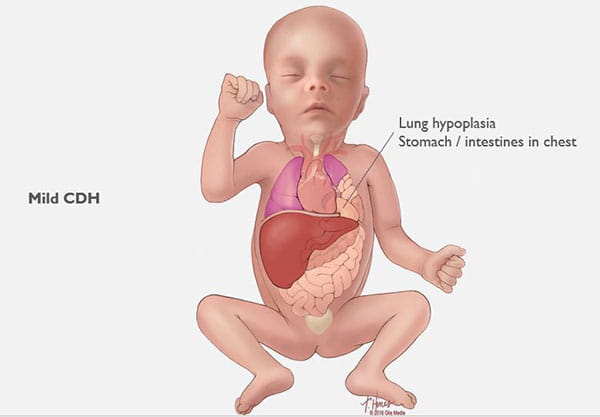
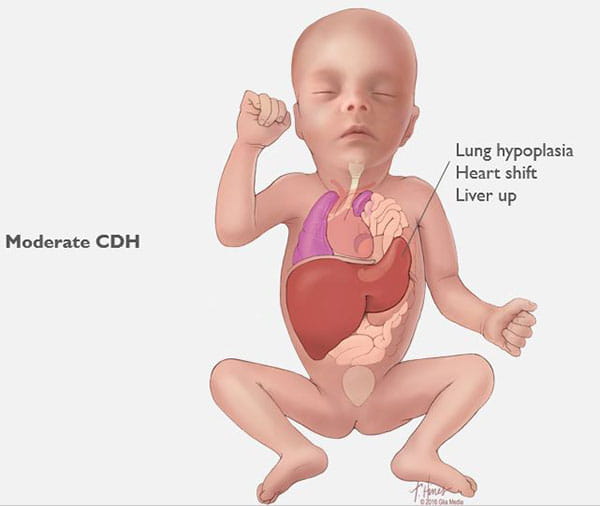
Congenital Diaphragmatic Hernia (CDH) Care at Cincinnati Children’s
Our Fetal Care Center specialists treat many fetal conditions once considered life-threatening or only treatable after birth. While many conditions can be managed through noninvasive therapies, some fetal conditions require surgery.
The Fetal Care Center is one of only a few fetal surgery centers in the U.S. with the expertise to perform the full range of fetal surgical interventions, including fetoscopic surgery.
Why Choose Us
Top-Ranked Center: Our neonatology program is ranked in the top 10 in the nation by U.S. News & World Report.
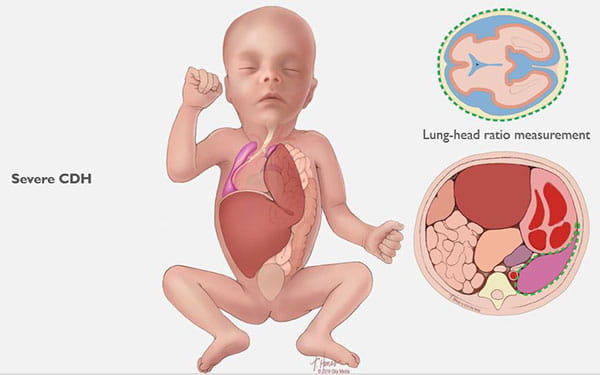
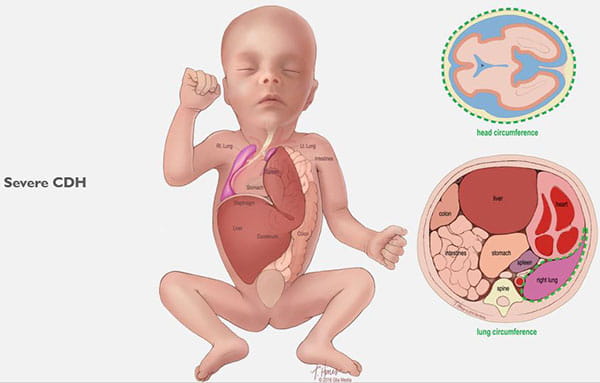
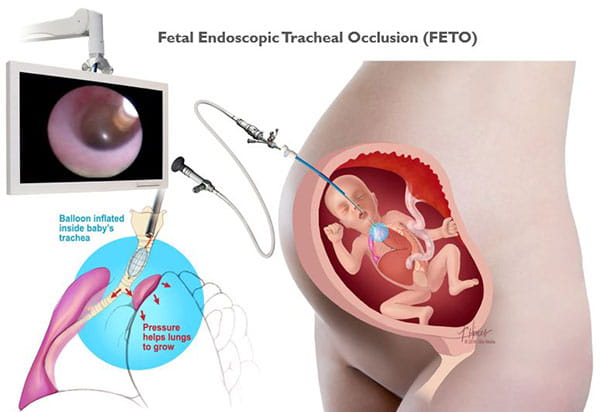
Deep Expertise and Experience: Our team specializes in treating complex and rare fetal conditions and is at the forefront of the innovation in fetal diagnosis and intervention therapies. Our surgeons have performed more than 50 Fetoscopic Endoluminal Tracheal Occlusion (FETO) procedures for congenital diaphragmatic hernia (CDH).
Dedicated CDH Team: Cincinnati Children’s has a dedicated program for children with CDH. Our team of CDH experts provide lifelong care and includes maternal-fetal medicine specialists, neonatologists, pediatric surgeons, pediatric cardiologists, ECMO specialists, nutritionists and therapists.
One-on-One Support: Through our family-centered approach to care, our nurse coordinators create a personalized program to support you and your baby at every turn. Throughout your time with us, you will be followed by a nurse coordinator who specializes in fetal care. Your nurse coordinator will be available to answer questions, as well as provide education and support for you and your family.