Referral Leads to Identification of New Genetic Lung Disease
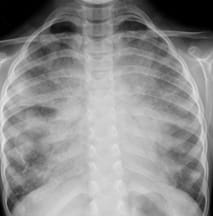
In October 2007, a 6-year-old girl with progressive dyspnea of several years’ duration was referred to Cincinnati Children’s for evaluation and treatment of pulmonary alveolar proteinosis (PAP). Previously diagnosed with asthma, she had failed standard treatments. Prompted by the severity of her dyspnea and an abnormal chest X-ray, a chest CT scan was performed and revealed diffuse bilateral ground glass opacification and septal thickening. Upon confirmation of PAP by surgical open lung biopsy, the child’s physician referred her to Cincinnati Children’s for evaluation of the etiology of PAP and for whole lung lavage therapy.
Our Approach: Integration of Clinical Care, Basic Science and Translational Research
Because of the patient’s unusual presentation, Bruce Trapnell, MD, director of Translational Pulmonary Medicine Research at Cincinnati Children’s and director of the Rare Lung Diseases Network, was brought in for consultation. This child’s lung disease represented none of the known disorders of surfactant homeostasis: autoimmune PAP (~90 percent of cases); secondary PAP (8 percent-9 percent of cases); or disorders of surfactant production (<1 percent-2 percent). Therefore, novel blood tests were developed in Trapnell’s research laboratory to evaluate this patient and confirmed the atypical nature of her disease, identifying abnormal GM-CSF receptor signaling as the cause. A series of detailed studies done on blood samples from the patient and members of her family identified inherited compound heterozygous mutations of the GM-CSF receptor alpha chain gene (CSF2RA) as the molecular etiology of PAP in this patient.
Clinical Outcome and Follow Up
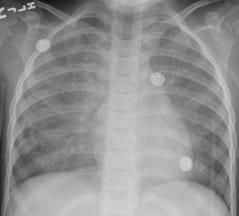
Robert E. Wood, PhD, MD, director of the Flexible Bronchoscopy Program at Cincinnati Children’s, performed the patient’s whole lung lavage therapy using techniques he developed specifically to perform this procedure in children. Bilateral whole lung lavage resulted in marked clinical, physiological and radiographic improvement.
Whole Lung Lavage Therapy |
|
|
|
Before whole lung lavage |
After whole lung lavage |
Two years after her treatment at Cincinnati Children’s, the patient’s exercise tolerance remains improved, and she is engaged in more normal daily activities. Based on the expected natural history of this disease, recurrence is likely and future treatments will be required.
Discussion and Lessons Learned: Discovery of a New Disease, Diagnostic Methods and Treatment Options
This case illustrates the importance of partnerships among clinicians and researchers in the care of children with rare, often poorly understood diseases. When this child’s lung disease could not be explained by state-of-the-art medical diagnostics, translational research studies were immediately initiated to define it at the molecular level.
“The translational research studies elucidated the molecular basis of PAP in this patient, simultaneously defining a new disease and novel diagnostic tests that we used subsequently to identify this disease among children throughout the world with unexplained PAP,” said Trapnell. “These are the first data to demonstrate familial PAP in humans and the critical role of the gene CSF2RA in pulmonary surfactant homeostasis.”
Basic research studies suggested the molecular defect in this child can be treated by inhaled GM-CSF therapy or bone marrow transplantation.