Experimental Drug Targets Nucleus of Allergen-Sensitized Cells
Experimental Drug Targets Nucleus of Allergen-Sensitized Cells
Study Suggests Blocking Transcription Factor to Treat Severe Lung Ailments
Tuesday, April 18, 2017
Transcription factors, the tiny proteins that switch genes on or off in the nucleus of cells, are considered unreachable molecular targets for drugs attempting to treat medical conditions. Overcoming this challenge, researchers at Cincinnati Children’s Hospital Medical Center discovered a small molecular compound that successfully blocks a transcription factor and its pro-inflammatory and hyper-mucous activity in asthma.
In a study published online April 18 by Science Signaling, scientists test a new compound they call RCM-1. The substance inhibits the transcription factor FOXM1 and a long downstream chain of pro-inflammatory processes it fuels in asthmatic airways. They show that RMC-1 treatment prevents overproduction of mucous-generating goblet cells in the lungs of mice with asthma and in human airway cell cultures.
Calling their compound a new therapeutic candidate for people with severe asthma and other chronic airway diseases, researchers say with additional research and development their discovery could lead to future clinical trials for asthma, Cystic Fibrosis and Chronic Obstructive Pulmonary Disease (COPD). All are severe pulmonary diseases associated with increased inflammation and mucous hyper-secretion.
“Traditional targets for drugs are receptors on cell surfaces, which are easy to reach. Transcription factors are inside cell nuclei and difficult to reach,” said Vladimir Kalinichenko, MD, PhD, the study’s lead investigator and a member of the Division of Pulmonary Biology. “RCM-1 keeps FOXM1 from entering the cell nucleus by activating cell machinery called proteasomes that degrade the transcription factor. This was very efficient at reducing lung inflammation and production of mucous-generating goblet cells in our tests.”
Better treatments needed
Kalinichenko said the research is driven by the need to develop more effective treatments for people with severe asthma and other debilitating lung ailments like COPD. Current clinical management for asthma focuses on reducing lung and airway inflammation triggered by allergens, such as house dust mites, mold, etc. Therapeutic agents able to directly affect gene regulatory networks that trigger inflammation and mucous-producing goblet cells would potentially be more efficient at slowing or stopping disease progression.
Leveraging the ongoing research of his laboratory into FOXM1 – which also plays a key role in lung cancer – Kalinichenko’s team identified the RCM-1 compound during a computer-supported biological imaging screen of 50,000 small compounds stored at the University of Cincinnati Genome Research Center. The scientists searched specifically for compounds that would target FOXM1 and inhibit its activation of downstream pro-inflammatory molecules that drive overproduction of mucous-generating goblet cells.
Successful lab testing
The research team then tested RMC-1 in cultured human airway epithelial cells and mouse models of asthma (via injection into the animals’ peritoneal cavities). The mice were either sensitized with house dust mite allergens or treated with a pro-inflammatory molecule called interleukin 13 (IL-13), which induces mucous production in airway diseases.
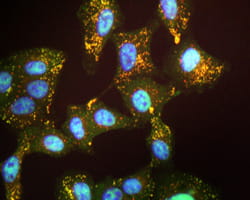
Microscopic images show that RMC-1 prevented FOXM1 from entering the nucleus in cultured human airway epithelial cells and in mouse allergen-sensitized respiratory airways. It also decreased airway hyper-responsiveness to allergen, reduced lung inflammation and improved lung function in mice sensitized to house dust mite allergens. FOXM1 also prevented IL-13 from causing goblet cell expansion in mice that were given the pro-inflammatory molecule intra-nasally.
Translating the science
Before the research team’s identification of RMC-1 could eventually lead to clinical trials, Kalinichenko said the researchers will first need to test RCM-1 in more sophisticated animal models of respiratory diseases. This will help address issues related to dosage, toxicity, optimal methods of therapeutic delivery, etc.
In their future work, the researchers also want to refine the chemical structure of RCM-1 to make it more efficient and to improve the method of therapeutic delivery. This includes seeing if the compound can be packaged into nanoparticles for intravenous injection. The researchers have applied for a patent on RCM-1, working through the Cincinnati Children’s Center for Technology Commercialization.
Funding support for this research came in part from: The National Institutes of Health (HL84151, HL123490, HL132849, CA142724); a Research Scholar Grant (RSG-13-325) from the American Cancer Society; the National Health Research Institutes of Taiwan (NHRI-EX105-10309BC); and from the Ministry of Science and Technology of Taiwan (MOST102-2628-B-007-003-MY3).