Daniel Lucas, PhD
- Member, Division of Experimental Hematology & Cancer Biology
- Associate Professor, UC Department of Pediatrics
About
Biography
I’m a scientist who studies hematopoiesis, bone marrow anatomy, bone marrow regeneration and leukemia. Since joining Cincinnati Children’s in 2018, I’ve focused on understanding the basis of blood cell production in the bone marrow.
As a PhD student at the Universidad Autonoma de Madrid in Spain, I generated mice lacking DNA polymerase mu (an enzyme involved in DNA repair). I found that these mice had a defect in their ability to produce blood. This discovery sparked my lifelong interest in regenerative/stress hematopoiesis and prompted me to continue my training at Mount Sinai Health System and Albert Einstein College of Medicine in New York. There, I worked with Paul S. Frenette, MD. His laboratory is interested in understanding how hematopoietic stem cells (HSCs) are regulated by the microenvironment of bone marrow.
During my postdoctoral training, I demonstrated that: 1) the sympathetic nervous system acts on the niche to control HSC trafficking and mobilization, and 2) sympathetic signals are necessary for the regeneration of the niche after transplantation. I also investigated the crosstalk between mature hematopoietic cells and HSC and discovered that macrophages and megakaryocytes are components of the niche regulating HSC trafficking and quiescence, respectively.
Today, as an independent principal investigator, I continue to study cellular crosstalk in the bone marrow and how it impacts hematopoiesis. My colleagues and I are also working to define the impact of different stromal niches in regulating steady state and emergency myeloid and erythroid cell production.
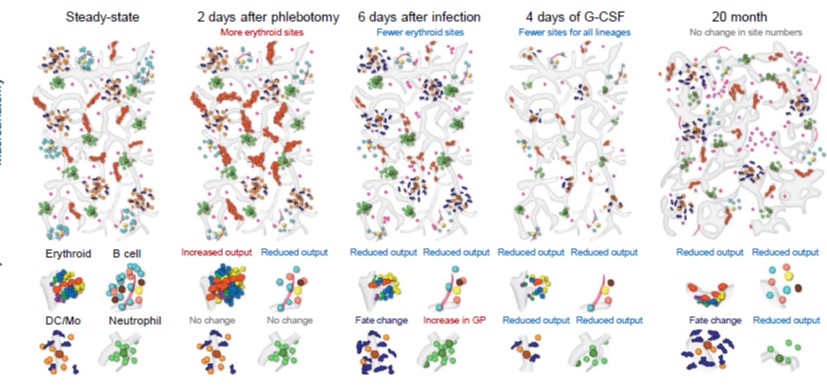
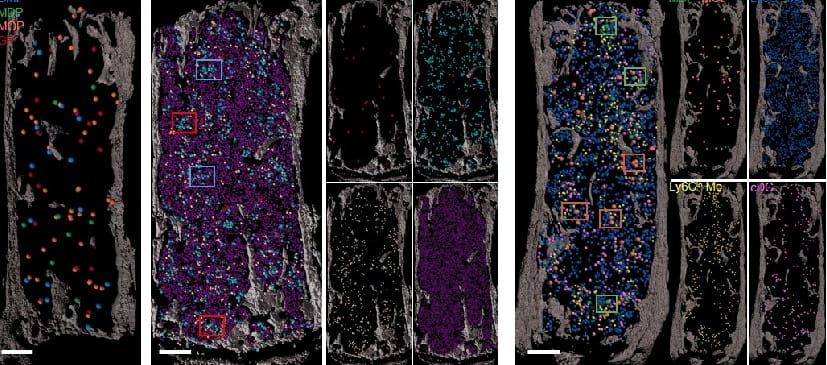
In addition to discovering that granulocytes drive the regeneration of the sinusoidal HSC niche after myeloablation via TNFα secretion, we recently developed a new series of imaging tools that allows complete mapping of myelopoiesis and erythropoiesis.
BS: Universidad Autonoma de Madrid, Madrid, Spain
PhD: Universidad Autonoma de Madrid, Madrid, Spain
Services and Specialties
Interests
Bone marrow; hematopoiesis; imaging; immunology
Research Areas
Publications
In situ mapping identifies distinct vascular niches for myelopoiesis. Nature. 2021; 590(7846):457-462.
A Tie2-Notch1 signaling axis regulates regeneration of the endothelial bone marrow niche. Haematologica. 2019; 104(11):2164-2177.
Hox11 expressing regional skeletal stem cells are progenitors for osteoblasts, chondrocytes and adipocytes throughout life. Nature Communications. 2019; 10(1):3168.
Neutrophils as regulators of the hematopoietic niche. Blood. 2019; 133(20):2140-2148.
Leukocyte Trafficking and Regulation of Murine Hematopoietic Stem Cells and Their Niches. Frontiers in Immunology. 2019; 10:387.
Dynamic regulation of hematopoietic stem cells by bone marrow niches. Current Stem Cell Reports. 2018; 4(3):201-208.
Granulocyte-derived TNFα promotes vascular and hematopoietic regeneration in the bone marrow. Nature Medicine. 2018; 24(1):95-102.
The orphan nuclear receptor TR4 regulates erythroid cell proliferation and maturation. Blood. 2017; 130(23):2537-2547.
Differential regulation of fetal bone marrow and liver hematopoiesis by yolk-sac-derived myeloid cells. Nature Communications. 2025; 16(1):4427.
Macrophage memories of early-life injury drive neonatal nociceptive priming. Cell reports. 2024; 43(5):114129.
From the Blog
First Skeleton-Wide Study of Blood Cell Formation Yields Surprising Findings
Daniel Lucas, PhD3/20/2024
Bone Marrow ‘Map’ Opens Path to Organoid-like Blood Stem Cell Production
Daniel Lucas, PhD, Nathan Salomonis, PhD ...2/10/2021