Juan Sanchez Gurmaches, PhD
- Assistant Professor, UC Department of Pediatrics
About
Biography
Obesity is one of the principal risk factors for metabolic dysregulations, including Type 2 diabetes, as well as cardiovascular disease and stroke. As a consequence, obesity creates incredible emotional, health and economic burdens worldwide. We are in desperate need of new therapies to reduce the effects of obesity.
The long-term goal of my laboratory is to identify new therapeutic approaches to treat or prevent obesity-derived pathologies by understanding the physiologic and pathologic fundamental mechanisms that control adipose tissue development, growth and metabolism.
In my lab, we use innovative lineage tracing techniques that allow single-cell resolution and a combination of whole-genome expression profiling, metabolomics, biochemical, physiological and cell biology approaches to find new therapeutic interventions against obesity.
Our findings indicate that adipose tissue, in contrast to other tissues and organs, carries a complexity of multiple stem cell and fully differentiated cell populations with complex dynamics and plasticity capabilities. Exploiting this developmental heterogeneity in the formation of adipose tissues, we demonstrated that genetically modifying the activity of the critical insulin/Akt/mTOR signaling pathway between adipocyte lineages during development significantly affects fat distribution, which, interestingly, is reminiscent of fat distribution observed in humans.
My research was featured on the cover of Cell Metabolism and Trends in Cell Biology, and several of my publications were selected for “best of” or “highlights” sections of their respective journals. In 2018, I was awarded with the Career Development Award by the American Heart Association and in 2020, with the Trustee Award from Cincinnati Children’s Hospital Research Foundation.
BS: University of Barcelona, Catalunya, Spain, 2005
PhD: University of Barcelona, Catalunya, Spain, 2010
Services and Specialties
Interests
Adipocytes; brown and white fat; animal models; adipocyte development and heterogeneity; insulin signaling; animal models
Publications
A shift in PKM2 oligomeric state instructs adipocyte inflammatory potential. JCI Insight. 2025; 10(22).
The widely used Ucp1-Cre transgene elicits complex developmental and metabolic phenotypes. Nature Communications. 2025; 16(1):770.
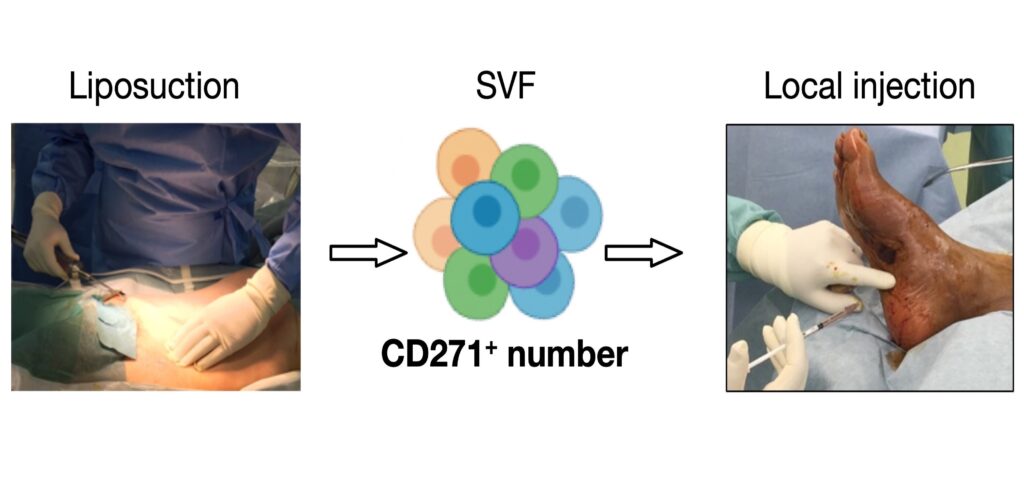
Single-cell transcriptomics identifies adipose tissue CD271+ progenitors for enhanced angiogenesis in limb ischemia. Cell Reports Medicine. 2023; 4(12):101337.
Quantitative Determination of De Novo Fatty Acid Synthesis in Brown Adipose Tissue Using Deuterium Oxide. Journal of Visualized Experiments. 2023; 195.
SEX-DEPENDENT EFFECTS OF ADIPOCYTE STAT3 INHIBITION ON THE INFLAMMATORY RESPONSE DURING SEVERE SEPSIS. Shock. 2023; 59(5):779-790.
Fluorescent Genetic Tools for Studying Brown Fat Development and Function in Mice. Methods in Molecular Biology. 2022; 2448:203-215.
A BAFF/APRIL axis regulates obesogenic diet-driven weight gain. Nature Communications. 2021; 12(1):2911.
The Lipid Handling Capacity of Subcutaneous Fat Is Programmed by mTORC2 during Development. Cell reports. 2020; 33(1):108223.
Violet-light suppression of thermogenesis by opsin 5 hypothalamic neurons. Nature. 2020; 585(7825):420-425.
Adaptive Thermogenesis in Mice Is Enhanced by Opsin 3-Dependent Adipocyte Light Sensing. Cell reports. 2020; 30(3):672-686.e8.
From the Blog
Novel Approach Emerging for Rescuing Limbs at Risk
Juan Sanchez Gurmaches, PhD12/19/2023