Cincinnati Children's scientists identify hormone that might help treat malabsorption
Cincinnati Children's scientists identify hormone that might help treat malabsorption
Human intestinal organoids grown from stem cells used to model congenital disorder in babies
Tuesday, September 22, 2020
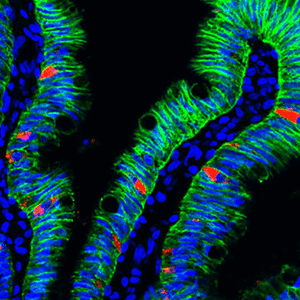
Scientists at Cincinnati Children's used human intestinal organoids grown from stem cells to discover how our bodies control the absorption of nutrients from the food we eat. They further found that one hormone might be able to reverse a congenital disorder in babies who cannot adequately absorb nutrients and need intravenous feeding to survive.
Heather A. McCauley, PhD, a research associate at Cincinnati Children's Hospital Medical Center, found that the hormone peptide YY, also called PYY, can reverse congenital malabsorption in mice. With a single PYY injection per day, 80% of the mice survived. Normally, only 20% to 30% survive.
This indicates PYY might be a possible therapeutic for people with severe malabsorption.
Poor absorption of macronutrients is a global health concern, underlying ailments such as malnutrition, intestinal infections and short-gut syndrome. So, identification of factors regulating nutrient absorption has significant therapeutic potential, the researchers noted.
McCauley was lead author of a manuscript published Sept. 22 in Nature Communications, which reported that the absorption of nutrients - in particular, carbohydrates and proteins - is controlled by enteroendocrine cells in the gastrointestinal tract.
Babies born without enteroendocrine cells - or whose enteroendocrine cells don't function properly - have severe malabsorption and require IV nutrition.
"This study allowed us to understand how important this one rare cell type is in controlling how the intestine absorbs nutrients and functions on a daily basis," McCauley said.
The Cincinnati Children's study, "Enteroendocrine cells couple nutrient sensing to nutrient absorption by regulating ion transport," was the first to describe a mechanism linking enteroendocrine cells to the absorption of macronutrients like carbohydrates and amino acids.
One key finding of the study is how these cells, upon sensing ingested nutrients, prepare the intestine to absorb nutrients by controlling the influx and outflux of electrolytes and water, the researchers stated. Absorption of carbohydrates and protein is then linked to the movement of ions in the intestine.
For this study, the scientists relied on human intestinal organoid models created in a lab, said James Wells, PhD, senior author of the study and chief scientific officer of the Center for Stem Cell and Organoid Medicine (CuSTOM) at Cincinnati Children's.
Grown from stem cells, organoids are small formations of human organ that have an architecture and functions that are similar to their full-size counterparts.
Cincinnati Children's launched efforts to make organoids from human pluripotent stem cells in 2006, said Wells, who is also director for basic research in the Division of Endocrinology at the medical center and an Allen Foundation Distinguished Investigator.
"What this study highlights is how decades of basic research into how organs are made and how they function is now leading to breakthroughs in identifying new therapeutics," said Wells, who has led a team of investigators at Cincinnati Children's who developed some of the first human organoid technologies that are now used globally.
The study on malabsorption used three different human small intestinal tissue models - all derived from pluripotent stem cells, which can form any kind of tissue in the body.
"The human organoids are essentially a much more realistic avatar to these patients with these rare mutations," Wells said. "They allow us to model much more faithfully the human disease."
McCauley and Wells conceived and initiated the recent study on malabsorption, designed the experiments and wrote the manuscript. Contributors to the study included intestinal physiology experts Marshall "Chip" Montrose, PhD, and Eitaro Aihara, PhD, of the University of Cincinnati.
About this Study
The study was supported by grants from the National Institutes of Health (U19 AI116491, P01 HD093363, UG3 DK119982, U01 DK103117); S&R Foundation and American Physiological Society; the American Diabetes Association (1-17-PDF-102); the Shipley Foundation and the Allen Foundation. Support was also received from the Digestive Disease Research Center (P30 DK078392).
Contact Information
Barrett J. Brunsman
barrett.brunsman@cchmc.org