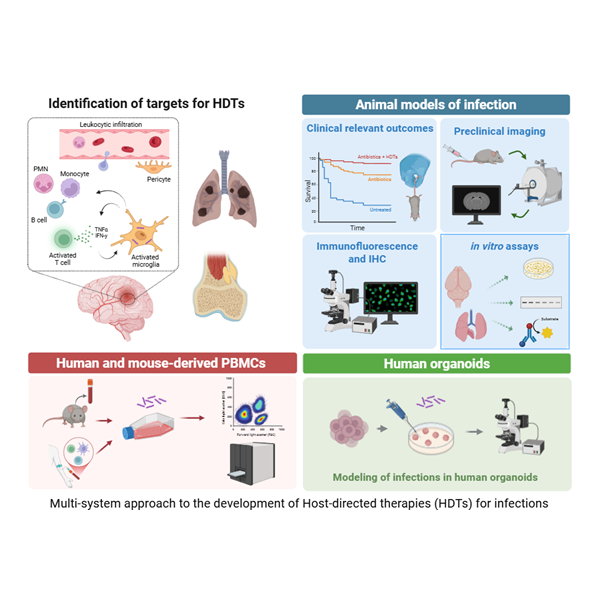
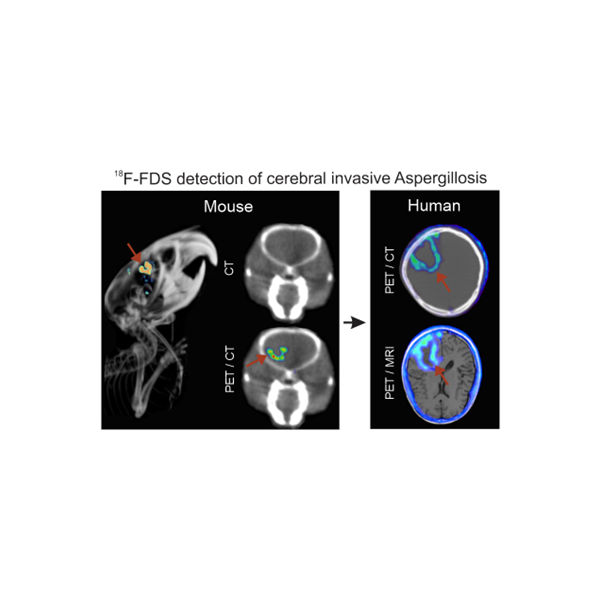
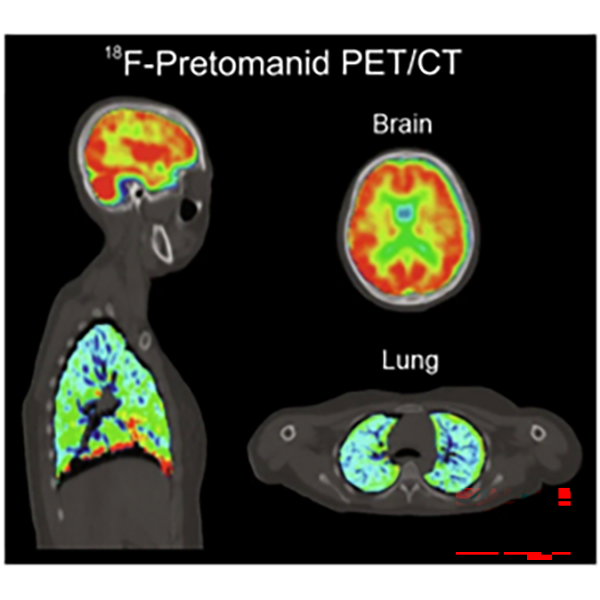
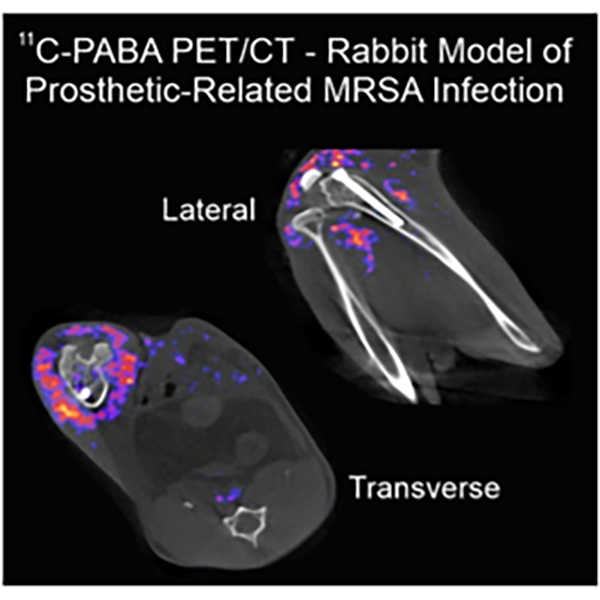
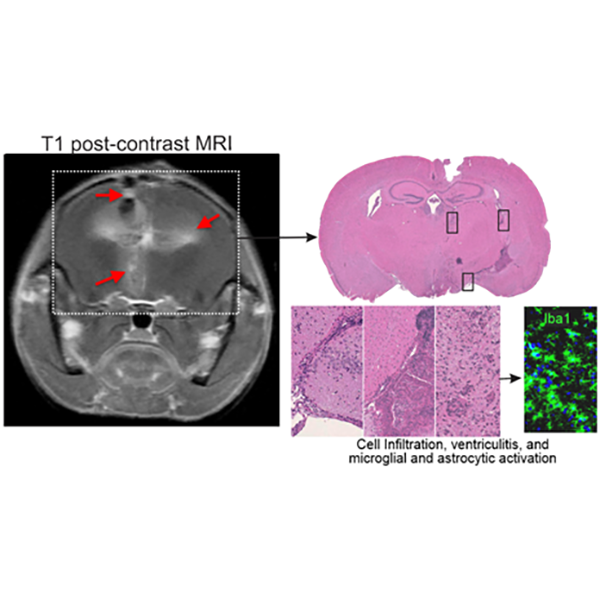
Recapitulation of the key features of human disease in animal models of tuberculosis is a critical step toward understanding pathophysiology and developing effective therapeutics. Using PET/CT, Dr. Jain demonstrated that C3HeB/FeJ (“Kramnik”) mice develop hypoxic TB lesions as well as cavitary disease—a fundamental discovery that reshaped the preclinical TB landscape and overturned the long-held dogma that matrix metalloproteinase-1 is the critical determinant of cavitation. “Kramnik” mice are now widely used in TB research worldwide, and new mechanisms underlying necrosis and cavitation continue to be explored. Similarly, a well-established model of TB meningitis has been developed in Dr. Jain’s laboratory that recapitulates key hallmarks of human disease. This model has been integrated with complementary platforms, including human brain organoids, primary human cell cultures, and advanced in vivo imaging. Using this multidisciplinary approach, we evaluate the biodistribution and penetration of TB drugs within infected brain tissues and optimize therapeutic regimens. In parallel, we investigate how intracerebral inflammatory responses and local antimicrobial exposure influence treatment efficacy, while also testing novel host-directed therapies (HDTs) that modulate neuroinflammation and reduce cerebral injury. By correlating drug distribution, host responses, and clinical outcomes, our goal is to inform the development of more effective, targeted therapies for this devastating form of tuberculosis.