About
Biography
My research areas include allergic and immune disorders. Proteases and protease inhibitors are well known for their important regulatory roles in the coagulation system, fibrinolysis and complement system. My work combines basic and translational research to investigate how proteolytic activity regulation and proteolytic activity can be regulated to reveal new therapeutic avenues.
In our lab, we investigate the function and regulation of proteases in host defense and the onset and propagation of inflammatory and allergic diseases. The function of proteases is irreversible and, therefore, highly regulated, particularly by protease inhibitors coupling to the proteases. Protease–protease inhibitor coupling limits the proteolytic activity and initiation of signaling pathways by the protease.
I focus on deciphering the role of proteases and their inhibitors in the epithelium as part of homeostatic and surveillance mechanisms. Loss of protease inhibitors and alterations of proteolytic activity leads to tissue damage and may exert a paramount signal in the development of inflammatory diseases. We aim to decipher the molecular mechanisms that disrupt the balance between proteases and protease inhibitors. Our goal is to develop strategies for controlling proteolytic activity as an approach to treating immune diseases.
A breakdown of immunologic tolerance appears to be a key feature of allergic diseases. The mechanisms underlying the break of tolerance are not well understood and involve a combination of environmental and genetic factors. The epithelium is the first line of defense against potential insults, providing a physical barrier between the host and the external environment. The importance of epithelial integrity in allergic disease development is supported by clinical observations of atopic or allergic march, which is the natural history or typical progression of allergic diseases that often begin early in life. These conditions include atopic dermatitis (eczema), food allergy, allergic rhinitis (hay fever) and asthma.
At a barrier breach, harmless antigens may encounter immune cells, potentially leading to the production of a danger signal and priming a break in immune tolerance and an allergic response. My lab investigates the mechanisms by which protease–protease inhibitor imbalance promotes barrier breaches and loss of immunologic tolerance in the development of allergic diseases.
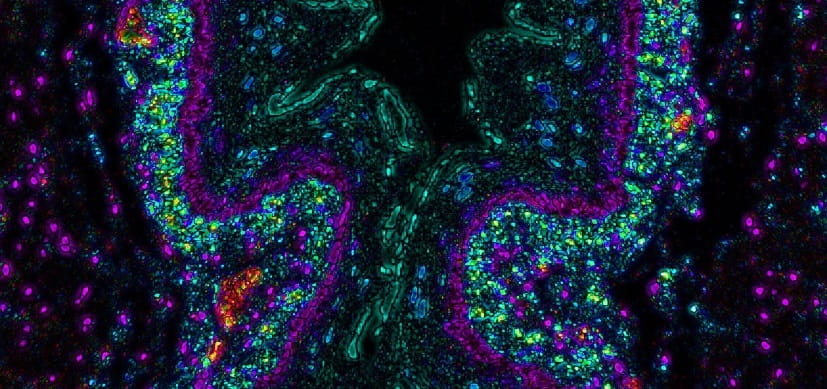
Some of the findings our lab discovered are that an imbalance of specific proteins, proteases and protease inhibitors in the cells lining the esophagus may cause inflammation and tissue damage in people with eosinophilic esophagitis (EoE). In people with active EoE, biopsies of esophageal tissues show a near-complete lack of the protease inhibitor SPINK7, which normally damps down inflammation and helps preserve tissue structure. In this study, researchers of the Azouz Lab and Rothenberg CURED Lab found a missing link that further explains how the process works. SPINK7 normally binds with the protease KLK5, keeping it in check. Without enough SPINK7, KLK5 is free to degrade tissues and initiate signals that lead to inflammation. These findings highlight esophageal allergy as a protease-mediated disease, suggesting that the disarming of proteases by delivering protease inhibitors can be therapeutic in allergic diseases.
I have more than eight years of research experience, and I began my work at Cincinnati Children’s in 2013.
PhD: Sackler School of Medicine, Tel-Aviv University, Israel, 2013
Msc: Sackler School of Medicine, Tel-Aviv University, Israel, 2008
Services and Specialties
Interests
Allergic diseases; the mechanisms by which protease–protease inhibitor imbalance promotes barrier breaches and loss of immunologic tolerance in the development of allergic diseases
Publications
The Bordetella type III secretion system effector BteA targets host eosinophil-epithelial signaling to promote IL-1Ra expression and persistence. Communications Biology. 2025; 8(1):1484.
Aryl hydrocarbon receptor and IL-13 signaling crosstalk in human keratinocytes and atopic dermatitis. Frontiers in Allergy. 2024; 5:1323405.
A novel class of TMPRSS2 inhibitors potently block SARS-CoV-2 and MERS-CoV viral entry and protect human epithelial lung cells. Proceedings of the National Academy of Sciences of the United States of America. 2021; 118(43).
Broad transcriptional response of the human esophageal epithelium to proton pump inhibitors. Journal of Allergy and Clinical Immunology. 2021; 147(5):1924-1935.
Molecular mechanism of inhibiting the SARS-CoV-2 cell entry facilitator TMPRSS2 with camostat and nafamostat. Chemical Science. 2021; 12(3):983-992.
Measurement of Exocytosis in Genetically Manipulated Mast Cells. Methods in Molecular Biology. 2021; 2233:181-192.
Alpha 1 Antitrypsin is an Inhibitor of the SARS-CoV-2-Priming Protease TMPRSS2. Pathogens and Immunity. 2021; 6(1):55-74.
Achieving Precision Medicine in Allergic Disease: Progress and Challenges. Frontiers in Immunology. 2021; 12:720746.
Uncovering the secretes of allergic inflammation. Journal of Clinical Investigation. 2020; 130(7):3419-3421.
Functional role of kallikrein 5 and proteinase-activated receptor 2 in eosinophilic esophagitis. Science Translational Medicine. 2020; 12(545).
From the Blog
FDA-Approved Drug Reverses Inflammation in Animal Model of Eosinophilic Esophagitis
Nurit Azouz, PhD, Marc E. Rothenberg, MD, PhD12/10/2020