Christina Gross, PhD
- Neuroscientist, Division of Neurology
- Associate Professor, UC Department of Pediatrics
About
Biography
To me, research is a joy and a passion, not just “a job.” I began my research career in 1999 because I wanted to learn more about how the brain works. To me, the brain is the most fascinating organ in the body, and I wanted to understand how we think, learn and feel. Later, I became more interested in how these mechanisms are disturbed in brain disorders.
It struck me that most brain disorders do not have good treatment options yet. Therefore, while I am still very interested in the basic brain mechanisms, I am now more dedicated to bridging the gap between brain disorders and treatments.
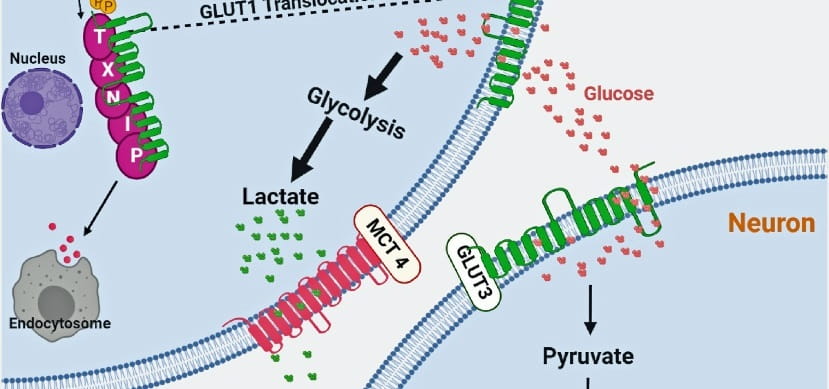
The overarching purpose of my research is to identify and understand pathological molecular mechanisms which contribute to autism and epilepsy. My lab works to discover how these mechanisms affect neuronal and brain function. Our ultimate goal is using this knowledge to identify molecular biomarkers and develop therapeutic strategies.
In more recent years, parts of my lab have tried to identify potential novel brain treatments. We discovered that a PI3K isoform-selective inhibitor ameliorates disease-relevant phenotypes in mouse models for two different neurodevelopmental brain disorders. We also discovered that a specific microRNA regulates neuronal hyperexcitability in mouse models of epilepsy.
My work has been funded by the National Institutes of Health (NIH) and other organizations, such as the Brain and Behavior Research Foundation, the American Epilepsy Society, and the Fragile X Syndrome Research and Treatment (FRAXA) Research Foundation.
Diploma: Biochemistry, Ruhr University Bochum, Germany, 2000
PhD: Free University of Berlin, Germany, 2004
Services and Specialties
Interests
Molecular neuroscience, fragile X syndrome
Research Areas
Publications
How Soon is Soon Enough? Consideration of Timing and Disease Etiology for microRNAs in Epilepsy. Epilepsy Currents. 2025; 25(6):410-415.
Altered PI3K/mTOR Signaling within the Forebrain Leads to Respiratory Deficits in a Mouse Model of Epilepsy. eNeuro. 2025; 12(12).
A companion to the development of common data elements for Sudden Unexpected Death in Epilepsy (SUDEP). Epilepsia Open. 2025; 10(5):1439-1449.
Enhancing sudden unexpected death in epilepsy (SUDEP) research through development of common data elements. Epilepsia Open. 2025; 10(5):1426-1438.
Somatic mosaicism and interneuron involvement in mTORopathies. Trends in Neurosciences. 2025; 48(5):362-376.
Beyond the Fragile X protein: neighborhood characteristics explain individual differences in IQ and adaptive behaviors of Fragile X syndrome. Frontiers in Psychiatry. 2025; 16:1636987.
Small Differences and Big Changes: The Many Variables of MicroRNA Expression and Function in the Brain. Journal of Neuroscience. 2024; 44(32).
Mir324 knockout regulates the structure of dendritic spines and impairs hippocampal long-term potentiation. Scientific Reports. 2023; 13(1):21919.
MicroRNA-218 instructs proper assembly of hippocampal networks. eLife. 2023; 12.
Age-Dependent Regulation of Dendritic Spine Density and Protein Expression in Mir324 KO Mice. Journal of Molecular Neuroscience. 2023; 73(9-10):818-830.
From the Blog
Scientists Discover Key Regulator of Neuron Function and Survival
Christina Gross, PhD9/1/2020