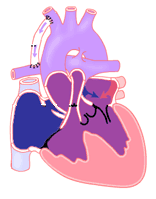
What is Tricuspid Atresia?
Tricuspid atresia is a type of congenital (a condition you are born with) heart disease. In this disease, the valve between the right atrium and right ventricle doesn’t develop.
Blood that returns from the body to the right atrium cannot directly enter the right ventricle. It must pass through a hole in the atrial septum (atrial septal defect) into the left atrium and then the left ventricle.
There are several differences that can produce different symptoms. The treatments depend on the defect and symptoms. There may be a hole in the ventricular septum, called a ventricular septal defect (VSD). The aorta and pulmonary artery may be normally positioned and in line with the appropriate ventricle (as shown in illustration). Or, they may be reversed, a condition called transposition of the great arteries. If the ventricular septal defect is small or absent, and the great arteries are normally positioned, blood flows from the left ventricle out the aorta to the body. In this situation, very little blood can get to the lungs. This leads to very low oxygen levels in the infant.
In a newborn baby, blood can reach the lungs to pick up oxygen if a connection between the aorta and pulmonary artery called the ductus arteriosus remains open. The "ductus" is an important vessel while the baby is still in the womb. It allows the blood from the baby's heart to return to the placenta. The placenta does the job of the lungs before birth. This vessel is sensitive to oxygen. When the baby is born, it closes completely after 24 to 48 hours. An intravenous medication called prostaglandin (PGE) can keep this vessel open after birth.
If a ventricular septal defect is present and the great arteries are in their normally related position, blood from the left ventricle can reach the lungs through the ventricular septal defect.
Finally, if there is transposition of the great arteries, blood reaches the lungs easily. This happens because the pulmonary artery is directly connected to the left ventricle. But blood can only reach the body and organs through the ductus arteriosus or the ventricular septal defect if there is one.
Signs and Symptoms of Tricuspid Atresia
The signs and symptoms of tricuspid atresia depend on the presence and size of the ventricular septal defect and the relationship of the great arteries.
Usually, the great arteries are normally related and there is either no ventricular septal defect or only a small ventricular septal defect. In this case, the newborn baby with tricuspid atresia will have a low oxygen level and a dusky, blue color, called cyanosis. This cyanosis may be noted at birth or it may be noticed after several days when the ductus arteriosus closes. As the ductus closes, the cyanosis worsens. If the cyanosis is severe enough, the baby will develop symptoms of poor oxygenation. This may be fast breathing or poor feeding.
Occasionally, there is an increased amount of blood flow to the lungs. This baby may have little to no cyanosis. They will likely develop congestive heart failure. Pulmonary edema (swelling in the lungs) can develop and cause the baby to breathe harder. The combination of increased heart and lung work uses large amounts of calories and results in congestive heart failure (CHF).
The symptoms of congestive heart failure are fast breathing, fast heart rate, sweating with feeds and poor weight gain. If there is transposition of the great arteries, the symptoms are related to low blood flow through the aorta and out to the body. This results in low blood pressure, fast breathing, poor feeding, cold, clammy hands and feet, and a pale, gray color. Infants with these symptoms are critically ill. They need emergency medical attention.
Diagnosis of Tricuspid Atresia
Babies with tricuspid atresia may or may not have a heart murmur. The amount of oxygen in the blood can be easily measured using a machine called a pulse oximeter.
Based on the symptoms reported by the parents and physical examination of the child, the doctor will order further tests to evaluate the heart.
The diagnosis of tricuspid atresia and the other problems such as a ventricular septal defect or transposition of the great arteries can be diagnosed by echocardiography. This involves using an ultrasound machine to make pictures of the heart. The pictures show the direction and amount of blood flow through the heart and blood vessels.
A chest X-ray and electrocardiogram (EKG) will often be done as well.
Treatment of Tricuspid Atresia
The diagnosis of tricuspid atresia with too little blood flow to the lungs or to the body needs immediate medical treatment. In a newborn baby (less than 1-2 weeks old), a medication called PGE can be given through an IV (intravenous line) to reopen the connection (ductus arteriosus) between the pulmonary artery and aorta. This improves blood flow to the lungs or body.
Children with tricuspid atresia and too little pulmonary blood flow will need surgery to create a connection between the arteries to the body and the arteries to the lungs. This type of surgery is called a modified Blalock-Taussig-Thomas (BTT) shunt. It involves the placement of a small tube between the artery to the arm (subclavian artery) and the arteries to the lungs (pulmonary artery). Sometimes a cardiac catheterization intervention is done to place a stent in the ductus arteriosus (PDA) instead of the surgical shunt.
If there is too much pulmonary blood flow (tricuspid atresia with a large ventricular septal defect), blood flow to the lungs will need to be limited. This is done to protect the lungs from becoming damaged. This can be done by placing a band around the pulmonary artery surgically or placing flow restrictors in the pulmonary arteries by a cardiac catheterization procedure so that blood flow to the lungs is limited but controlled.
Finally, if the problem is not enough blood flow through the aorta (tricuspid atresia with ventricular septal defect and transposition of the great arteries), blood from the normal-size left ventricle will need to be routed to the aorta. The aorta will need to be reconstructed. Pulmonary blood flow can be established by placement of a modified BTT shunt.
Whatever surgery is needed in the newborn period, children can expect to have further heart surgery by the age of 3-6 months.
The operation at 3-6 months is called a bidirectional Glenn. The superior vena cava is removed from the heart and connected directly to the pulmonary artery. The BTT shunt is removed. This allows blood from the upper body to flow directly to the lungs to pick up oxygen without having to be pumped by the heart. It also prevents blood that has oxygen from returning to the lungs. It keeps the heart from doing unnecessary work. After this surgery there is still blood returning from the body through the inferior vena cava going directly back to the body without first passing through the lungs. Because of this, some level of cyanosis will be present.
Between the ages of 2 and 5 years, children with tricuspid atresia will be ready for the third surgery. This surgery is called the Fontan procedure. It involves connection of the inferior vena cava to the pulmonary artery. This forces all blood returning from the body to pass through the lungs and pick up oxygen before being pumped to the body. This allows a more normal color in the skin and lips due to a more normal oxygen saturation in the blood.
Treatment of Tricuspid Atresia: Results
The results of this staged approach for children with tricuspid atresia are good. The expected survival through all three stages is 75-95% depending on the exact defects and surgery for any given person.
The long-term quality of life following the Fontan operation is good. The oldest patients are currently in their 30s. However, several complications may occur. These include heart rhythm problems needing medication or placement of a pacemaker. Patients may also have lung problems leading to a decrease in oxygen in the blood. Digestive problems caused by loss of protein in the stool may be a concern. This may lead to swelling and water retention. For some patients, heart and lung transplantation may be needed later in life.