Understanding the Developmental and Metabolic Basis of Adipocyte Heterogeneity
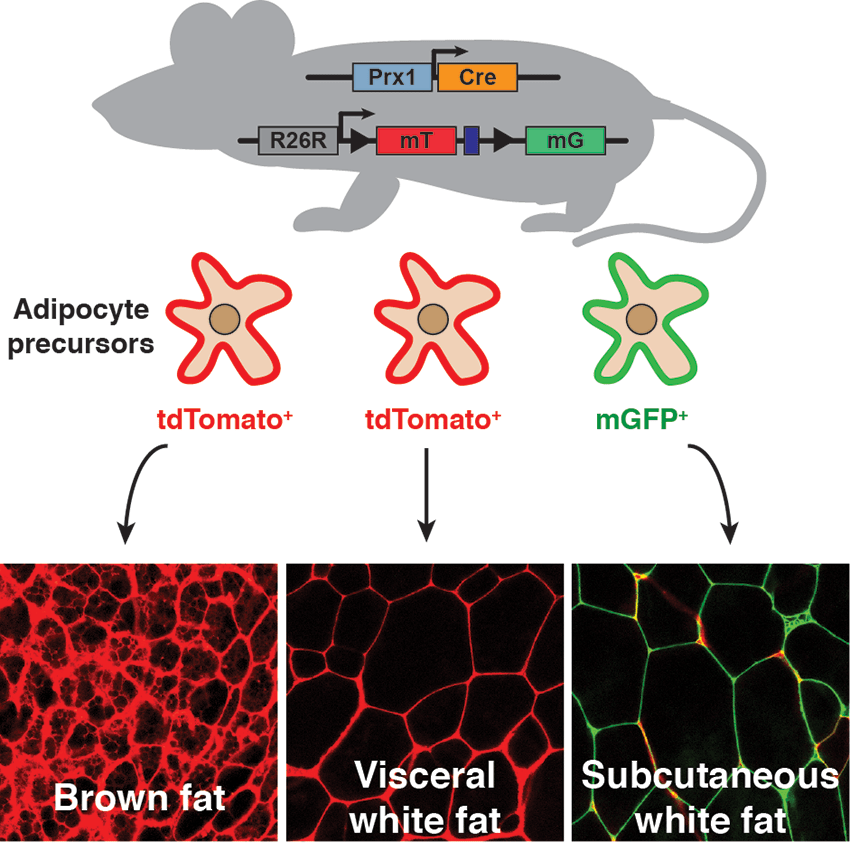
Adipose tissue shows a dramatic functional heterogeneity between depots (for example subcutaneous and visceral fat) but also between cohabitating adipocytes within a single depot. However, the mechanisms leading to this heterogeneity are not well understood. We have employed mouse genetics to partially identify the origins of adipocyte heterogeneity. This is thanks to innovative lineage tracing techniques that allow for single cell resolution at the adipocyte precursor and mature adipocyte levels using a Cre-sensitive dual-color cell membrane-labeling reporter. Using this approach with candidate Cre-drivers, we (1) showed that both brown and white adipocyte precursor and mature adipocytes are heterogeneous in its developmental origin, among and within fat depots; (2) reveled that the healthy subcutaneous white adipocytes originate from a distinct lineage than the unhealthy visceral white adipocytes (3) found evidence of lineage dynamics and plasticity including sex-linked, and diet-induced changes in lineage distribution. This body of work has delineated a new conceptual framework in our understanding of adipose tissue development and function that has been incorporated and expanded by the field, becoming an intense area of research.
We continue our effort developing this project in multiple directions: (1) using new mouse models for lineage specific cell isolation; (2) using whole tissue and single cell technologies together with CRISPR based cell labelling to map the developmental relationship between each fat pad and adipocytes within fat pads.