Rac GTPases in Erythropoiesis
Rac1 and Rac2 GTPases mediate proliferation, survival, adhesion and migration of hematopoietic stem cells and early myeloid and erythroid progenitors in the bone marrow microenvironment. Their deficiency results in massive mobilization of progenitors from the bone marrow and increased homing in the spleen, while it also disturbs the actin cytoskeleton of mature RBCs.
Our findings indicate that Rac1 and Rac2 GTPases are essential for normal bone marrow erythropoiesis; however they are dispensable for erythropoiesis in the spleen, implying different signaling pathways for homeostatic and stress erythropoiesis (Kalfa et al, Haematologica. Jan;95(1):27-35. 2010).
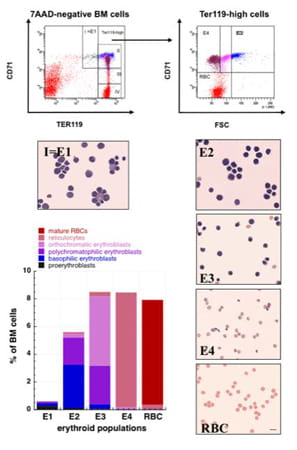
To assess erythroblast differentiation in the bone marrow and spleen of Rac1-/-;Rac2-/- versus wild-type mice, we re-evaluated a widely used flow-cytometry assay based on immunostaining for CD71 and Ter119. We observed that under our conditions of staining, using excess anti-CD71 antibody with the purpose of saturating the CD71 receptor and generating reproducible results, the population II became the major population of the Ter119+ cells.
We, therefore, proceeded to flow-sorting and morphological identification of the cells in the E1, E2, E3, E4 and R populations as shown. In agreement with previous studies in which transferrin receptors were quantified during erythroid development, the intensity of staining of basophilic, polychromatophilic and orthochromatic erythroblasts with anti-CD71 is similar, ranging within approximately one logarithmic scale and, therefore, these erythroblasts overlap within population II of the CD71-Ter119 cascade. The CD71-Ter119-high cells (population IV in the CD71 versus Ter119 flow cytogram equivalent to population R in the CD71 versus forward scatter plot) are more than 95 percent mature red blood cells (Kalfa et al, Haematologica. Jan;95(1):27-35. 2010).