Fetoscopic Endoluminal Tracheal Occlusion (FETO) for CDH
What is FETO?
Fetoscopic endoluminal tracheal occlusion, or FETO, is a surgical procedure to treat the most severe cases of congenital diaphragmatic hernia (CDH) that are diagnosed in utero. If you are pregnant and your baby has been diagnosed with CDH, you might be a candidate for the FETO procedure.
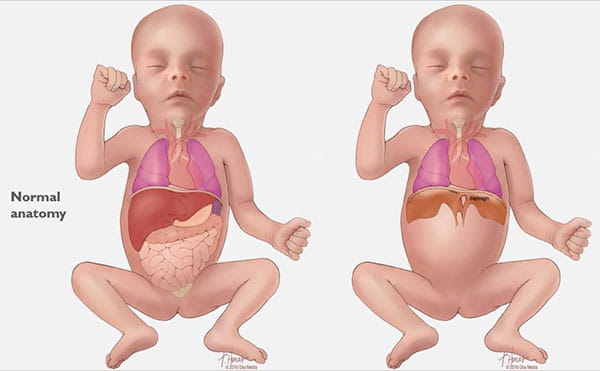
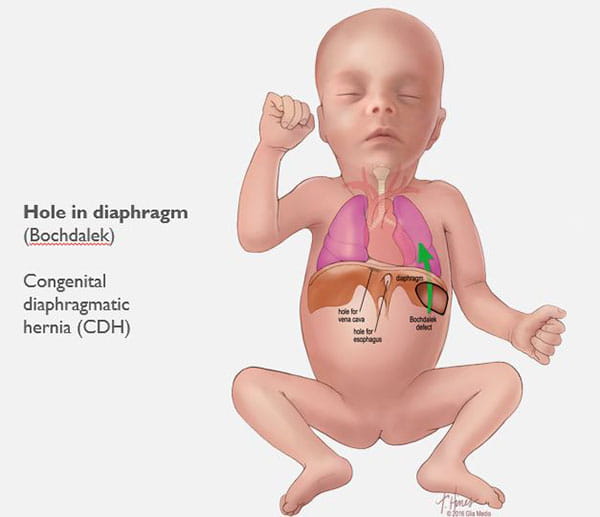
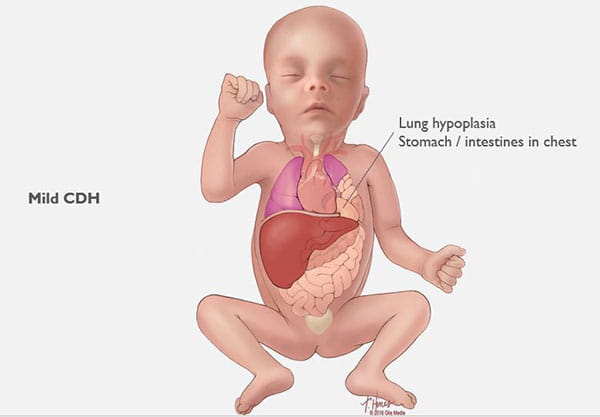
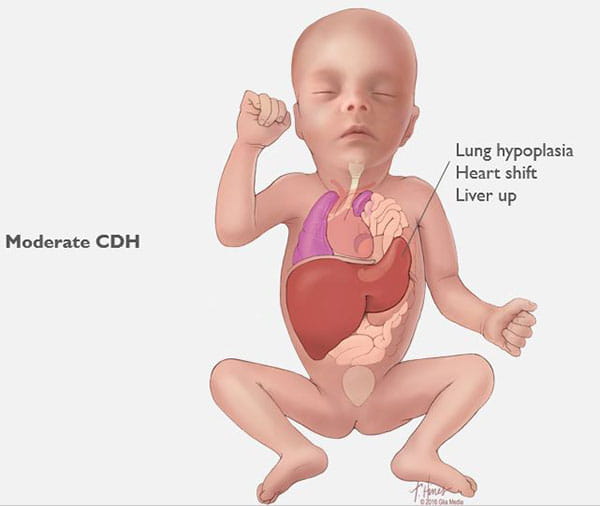
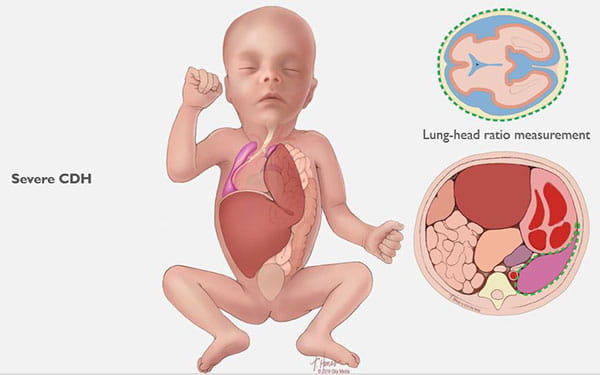
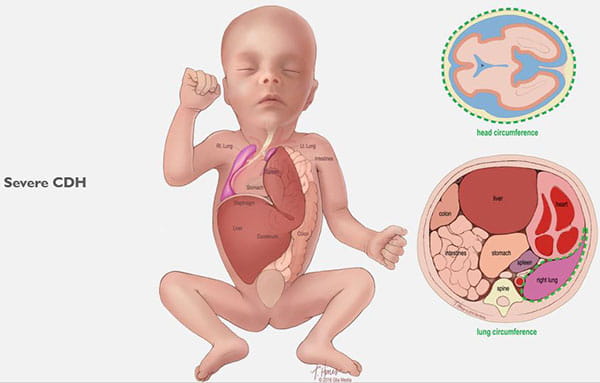
CDH occurs when a hole develops in your unborn baby’s diaphragm (the breathing muscle that separates the chest from the abdomen). The opening allows the abdominal organs to push into the baby’s chest and crowd the developing lungs. This can cause the lungs to remain small and underdeveloped. (See Images 1-6.)
CDH can lead to serious disease and can be life-threatening. In the most severe cases, treatment during pregnancy can allow the lungs to develop more fully and can increase the baby’s chance of survival.
What happens during a FETO procedure?
Normally a fetus’s lungs produce fluid that escapes through the trachea (windpipe). Blocking the trachea prevents this fluid from escaping, which can increase pressure in the trachea. Research has shown that building up this tracheal pressure and fluid content can stimulate lung growth. The FETO procedure provides an artificial way to block your baby’s trachea and promote lung development. (In a typically developing fetus, lung growth occurs naturally, so the trachea doesn’t need to be blocked.)
In a FETO procedure, between 27-31 weeks gestation, a small incision is made in the mother’s belly and uterus. An instrument is guided through the incision and into your baby’s trachea. Through a surgical instrument, a balloon is then inserted into the trachea and inflated with fluid to block (occlude) the trachea. The placenta provides oxygen for your baby, so the balloon doesn’t interfere with fetal breathing before birth. Click here to see a video of the balloon inflation.
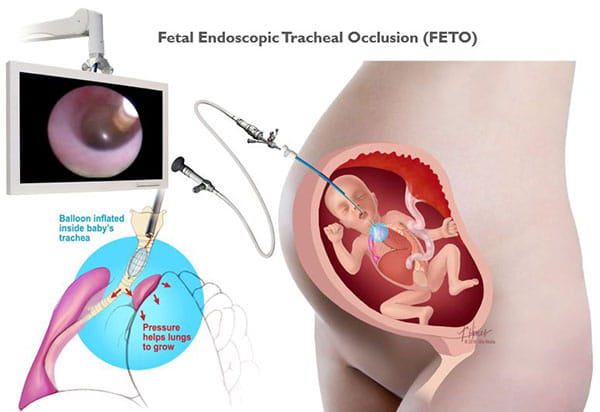
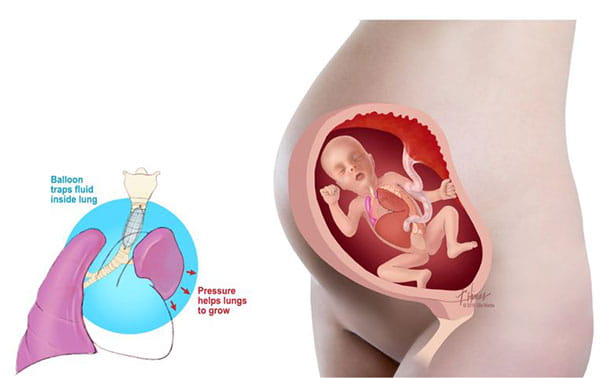
The balloon remains in your baby’s trachea for 4-6 weeks, until 32-34 weeks gestation. You will undergo regular ultrasounds during this time to monitor your baby’s condition.
A second procedure is performed to deflate or remove the balloon so your baby’s lungs can continue to mature. Once your baby is born, neonatal care will be provided and surgery will be performed to close the hole in the diaphragm.
Who can have the FETO procedure?
FETO is an experimental procedure. It is not an appropriate treatment for all cases of CDH diagnosed in utero. It is only offered in instances of severe CDH where we feel the mother and baby will have the most potential to benefit from the procedure. We conduct thorough diagnostics and review of a mother’s medical history to determine the appropriate treatment for our patients on an individual basis.
FETO evaluation
To determine if you are a candidate for the FETO procedure, you will have a standard prenatal evaluation at the Cincinnati Children's Fetal Care Center, which consists of the following:
• An ultrasound
• A fetal MRI
• A fetal echocardiogram to test your baby’s heart function
• A meeting with a nurse, social worker and genetic counselor
• A team meeting with a maternal-fetal medicine specialist (MFM), pediatric surgeon and neonatologist
After your tests are complete, our team of experts meets with you to discuss whether the FETO procedure is appropriate for you, risks to both mom and baby, and survival rates. We also discuss how severe your baby’s condition is, its impact on the remainder of the pregnancy, medical treatments that might be needed immediately following the birth of your child, and long-term prognosis of babies with CDH.
You do not have to choose to have the FETO procedure performed, even if you are a candidate for it.
What can I expect after the procedure?
If you are a candidate for and choose to have the FETO surgery, the procedure will be done at Cincinnati Children’s Hospital. You will need to remain in Cincinnati after the procedure and until your baby is born. This is because your baby’s airway is blocked temporarily, and unexpected delivery could be life-threatening without proper intensive care.
What will happen after my baby is born?
The plan for delivery and afterward is as follows:
• Mother delivers by C-section at Cincinnati Children’s Hospital with ECMO standby.
• Baby is transferred to the Cincinnati Children’s neonatal intensive care unit (NICU).
• Once stabilized, the baby undergoes surgery to repair the hole in the diaphragm.
Length of stay in the NICU depends on the baby’s pulmonary capacity, the recovery from surgery, how early the mother delivers, and any complications related to prematurity.
Volumes and Outcomes
Our Fetal Care Center surgeons have performed more than 50 FETO procedures.
Patients with severe CDH receiving FETO had lower rates of ECMO use, shorter days on mechanical ventilation, and shorter length of stay in the NICU.
Data based on patients cared for at the Cincinnati Children's Fetal Care Center Feb. 1, 2004, through June 30, 2023.