Genetic Basis and Pathogenic Mechanisms of Cleft Lip and Palate
Cleft lip and cleft palate (CLP) are the most common craniofacial birth defects, with a frequency of one in 300 to 2,500 infants, depending on population. Since the developmental processes underlying lip and palate morphogenesis are remarkably similar in mice and humans, the laboratory mouse provides an excellent model to understand both the normal developmental processes as well as pathogenic mechanisms of CLP.
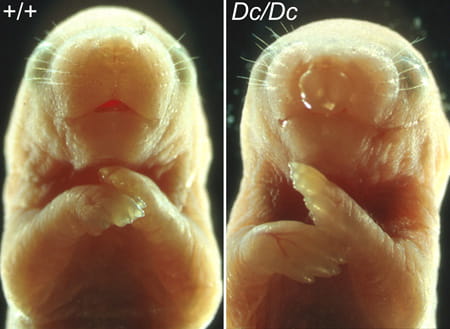
Previous studies identified several spontaneous mutant mouse strains with CLP phenotypes. Through high-resolution genetic mapping and genomic sequencing, we have identified the genetic basis of CLP phenotypes in the Dancer (Dc) and Twirler (Tw) mutant mice. Dc and Tw mutations each cause inner ear defects in heterozygous and CLP in homozygous mutant mice. We found that Dc resulted from an insertion mutation in the Tbx10 gene that encodes a T-box containing DNA-binding transcription factor. Tw, on the other hand, is caused by a single nucleotide change in the first intron of the Zeb1 gene, which encodes a homeodomain and zinc finger-containing transcription factor.
Ongoing research is investigating the cellular and molecular mechanisms underlying the CLP pathogenesis in these and other mutant mouse strains using a combination of gene expression profiling and molecular marker studies, as well as generation and analyses of tissue-specific transgenic mice.
In addition, we are using ENU mutagenesis screens to identify genes and gene-gene interactions underlying craniofacial birth defects. Several ENU-induced mutant mouse strains are currently being characterized to identify the causal genes and pathogenic mechanisms.