Neurofibroma Formation and Growth: The role of immune cells
The Ratner team found that 30% of neurofibroma cells are a type of white blood cell called a macrophage. Dr. Ratner’s team sought to use in vivo methods to test the relevancy of macrophages in the growth, development, and possible therapeutic resistance of neurofibromas. Initially, macrophages limit tumor growth. As a tumor becomes established, macrophages release cytokines and chemokines, which stimulate blood vessel growth (increasing tumor volume) and proteases, which allows the tumor to infiltrate tissues more easily. This stage-dependent role of macrophages indicates that macrophage inhibitors could represent a therapeutic strategy for established neurofibromas.
SC and macrophage gene expression data and computational reconstruction of molecular networks and signaling revealed inflammation pathways, predicted the changes of cytokines, chemokines, and growth factors. These predictions verified the presence of the macrophage chemo-attractant, colony stimulation factor 1, and predicted a role for type-1 interferon (IFN), a cytokine upstream in immune response signaling. This work provides a platform for future investigations to target neurofibroma therapy. Ongoing areas of research include the examination of the genetic makeup of macrophages and SC and the signals influencing potential cross-talk for Ras and interferon pathways.
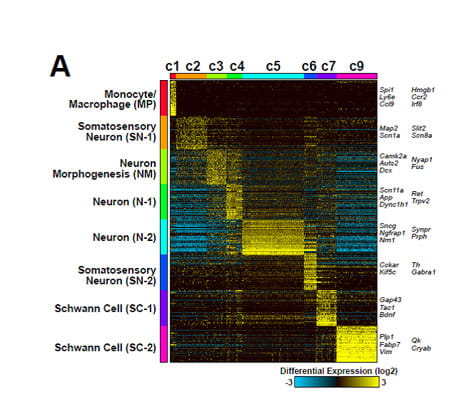
Jonathan Fletcher, a PSTP student in the laboratory, moved this project forward by characterizing immune cell populations, and investigating earlier stages of tumor formation using single cell RNA sequencing (with Nathan Salomonis). One of his studies identified Cxcl10/IP10 as a critical driver of neurofibroma formation. CXCL10 recruited from a subpopulation of Nf1-/- Schwann cells recruits other immune cells, driving tumor formation.