Hepatocyte Plasticity
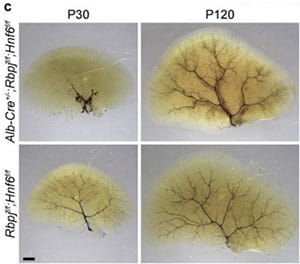
Transdifferentiation is a complete and stable change in cell identity that serves as an alternative to stem-cell-mediated organ regeneration. In adult mammals, findings of transdifferentiation have been limited to the replenishment of cells lost from preexisting structures, in the presence of a fully developed scaffold and niche. We have shown that transdifferentiation of hepatocytes in the mouse liver can build a structure that failed to form in development—the biliary system in a mouse model that mimics the hepatic phenotype of human Alagille syndrome (ALGS). In these mice, hepatocytes convert into mature cholangiocytes and form bile ducts that are effective in draining bile and persist after the cholestatic liver injury is reversed, consistent with transdifferentiation. These findings redefine hepatocyte plasticity, which appeared to be limited to metaplasia, defined as incomplete and transient biliary differentiation as an adaptation to cell injury.
To continue this investigation, we are focusing on 1) identification of the transdifferentiation-inducing signals, and 2) determination of the key transcription factors and targets required to reprogram hepatocytes to cholangiocytes. These avenues will reveal opportunities for the treatment of ALGS and other cholestatic liver diseases.