Current Projects
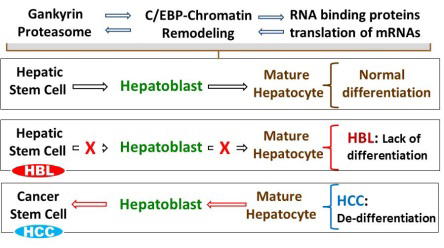
Timchenko's lab is working with three independent NIH-funded projects which investigate mechanisms of liver cancer. The united hypothesis of these projects is that disorganization of a network between proteasome, C/EBP-chromatin remodeling complexes and RNA binding proteins leads to elevation of Hepatic Stem Cells and CSCs and subsequent development of HCC and HBL (see Fig. 1). During normal post-natal development of the liver, the network of these three signaling pathways supports differentiation of hepatic stem cells into hepatoblasts and mature hepatocytes. In adult organisms, this network controls differentiation status of hepatocytes. The hypothesis is that HBL is caused by the failure of hepatic stem cells to differentiate into hepatocytes, while HCC is the result of de-differentiation of hepatocytes into CSCs with ensuing proliferation leading to liver cancer. This hypothesis is based on four major discoveries which have been made in Dr. Timchenko's lab over the last two years.
- First: A normal liver expresses several tumor suppressor proteins which protect the liver from development of cancer. Dr. Timchenko’s lab found that a small subunit of proteasome, gankyrin, is a key molecule which is activated during development of liver cancer and eliminates 5 tumor suppressor proteins including p53, Rb, C/EBP, HNF4 and p16. These findings have been published in Journal of Clinical Investigation and in Hepatology (Wang et al. JCI 2010; Jiang et al. Hepatology 2013) and are summarized in a recent review (Lewis & Timchenko, Cancer Studies and Molecular Medicine 2015).
- Second: The main characteristic of liver cancer is a failure of hepatocytes to stop proliferation leading to liver tumors. Using unique, genetically modified animal models, Dr. Timchenko’s lab found that a complex communication of C/EBP transcription factors and chromatin remodeling proteins is required for termination of liver proliferation. His recent work shows that disruption of this network leads to failure of liver to stop proliferation and potentially might lead to development of liver cancer. This finding has been recently published in Hepatology(Jin et al 2015).
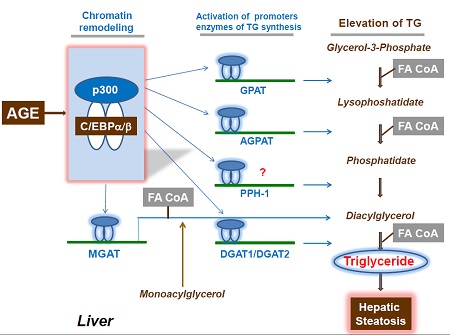
- Third: Dr. Timchenko’s lab investigates the mechanisms of NAFLD. NAFLD is a significant health problem which affects 30% of adult people. The earliest step of NAFLD is hepatic steatosis, which is developed in aging liver and under conditions of high fat diet. Timchenko’s lab has elucidated a molecular scenario of liver steatosis (see Fig. 2).
Timchenko’s lab has shown that high fat diet and age cause alterations of chromatin structure which are associated with accumulation of high MW complexes of C/EBP family proteins and p300. These complexes activate promoters of five enzymes of triglyceride synthesis leading to elevation of triglycerides and subsequent development of hepatic steatosis and NASH. These discoveries have been published in two recent papers (Jin et al, Cell Reports 2013; Breaux et al, Mol Cell Biol 2015). Dr. Timchenko’s lab is now working on the development of approaches for prevention and treatments of NAFLD. - Fourth: In collaboration with scientists at Cincinnati Children’s Hospital Medical Center, Dr. Timchenko started investigations of Bio Bank liver samples available at Cincinnati Children's. The initial studies with HBL samples revealed that Gankyrin-proteasome pathway is activated in hepatoblastoma samples. These observations support the main hypothesis of his lab and provide a background for development of Gankyrin-based approaches to treat liver cancer.