Tracheal Patterning
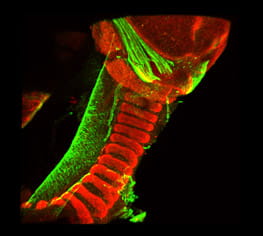
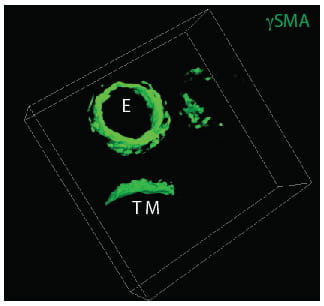
The mammalian trachea is patterned across the dorsal-ventral axis. Smooth muscle is present in the trachea's dorsal aspect, while C shape cartilaginous rings are located in the ventral side. This arrangement allows flexibility during inspiration and prevents the collapse of the walls of the trachea during expiration. It is unknown how the tracheal pattern is established; moreover, the source of the progenitor cells that give rise to tracheal muscle or cartilage has not been identified. Disrupted tracheal patterning is observed in congenital disabilities such as airway malacia. In tracheobronchomalacia (TBM), the trachea and bronchi walls are soft because of the lack of cartilage. Other conditions affecting the tracheal cartilage and muscle, such as complete tracheal rings (CTR) or tracheal sleeves, are also the result of abnormal tracheal patterning. While the conditions are associated with high morbidity, the causes of TBM and CTR in human patients are poorly understood. Our goal is to define the molecular mechanisms underlying the differentiation and organization of tracheal chondrocytes and myocytes.
At the molecular level, our laboratory focuses on the Wnt signaling pathway and its influence on the cross-talk between epithelium and mesenchyme of the trachea necessary for cell differentiation. Using mouse models, we are studying how the trachea's cartilage and muscle are specified and how abnormal Wnt signaling interacting with other signaling pathways affects tracheal cell differentiation and leads to tracheal malformations. In collaboration with physicians and researchers of Pulmonary Medicine and Perinatal Institute, we investigate genetic causes of conducting airway malformations in the pediatric population. Thus, the investigations aim to determine the causes of diseases affecting the trachea and bronchi's supporting structure by comparing mouse and human studies.
This work is supported by a grant from the NIH-NHLBI.