Spermatogonial Differentiation
One of the most important biological functions of the adult testis is to maintain fertility over an extended reproductive lifespan via the constant production of sperm. To produce millions of sperm daily, renewal and differentiation divisions are tightly regulated in the spermatogonial stem cell (SSC) population of the mammalian testis. Defects in either self-renewal or differentiation functions of SSCs lead to depletion of sperm and result in infertility.
The microenvironment that regulates stem cell renewal and differentiation divisions is called the stem cell niche. The germline stem cell niches of invertebrate model organisms have been well-defined, but the SSC niche is mammals has proven difficult to characterize. Recent evidence in the field suggests that signals from the testis interstitial compartment are important for function of the SSC niche. One interstitial cell type that has not been examined in this context is the testicular macrophage. Our recent results have implicated a role for macrophages in the differentiation of spermatogonia. We are interested in defining the signaling pathways used by macrophages to regulate testis function. We anticipate that defining these cellular relationships will lend insights into the process of spermatogenesis and help us understand the etiology of male infertility.
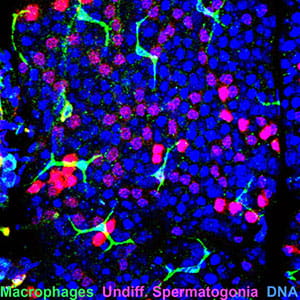
In a wild-type adult testis, macrophages are present on the surface of the seminiferous tubule, overlying the basement membrane and seminiferous epithelium. Numerous chains of undifferentiated spermatogonia are visible along the length of the tubule, just beneath peritubular macrophages.



