Fetal Gonad Morphogenesis
Prior to sex determination, XY (male) and XX (female) gonads are molecularly and morphologically indistinguishable from one another. Upon the expression of the Y-linked gene Sry in XY gonads, a cascade of genetic and cellular events takes place to initiate the testis morphogenesis program. Among these changes include: the specification of Sertoli cells, the somatic supporting lineage of the testis; the corralling of germ cells and Sertoli cells into testis cords, the precursors to adult seminiferous tubules; and the formation of the testis-specific vascular system. In the absence of Sry, XX gonads undergo the ovarian differentiation program.
While Sertoli cells are thought to be the central cell type driving testis morphogenesis, our previous results show that other cell types, such as interstitial cells and endothelial cells, are required to form testicular architecture. One cell type that is poorly understood in the context of fetal gonad development is the myeloid immune cell lineage, which includes macrophages. While they are traditionally known for their phagocytic roles in innate immunity, myeloid cells are also involved in vascular remodeling and tissue patterning during development. Our preliminary results indicate that macrophages play diverse and essential roles in testis differentiation. We seek to understand how macrophages interact with other cell types to promote testicular and ovarian architecture necessary for spermatogenesis and oogenesis, respectively, throughout life. We are also investigating how immune cells attain sexually dimorphic roles and phenotypes during gonad development.
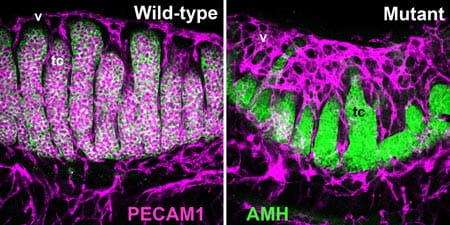
The E13.5 wild-type XY gonad shows well-developed testis cords (tc) and testis-specific coelomic vasculature (v). The presence of supernumerary macrophages in a macrophage differentiation mutant XY gonad is correlated with testis cord abnormalities, germ cell deficits, and hypervascularization. PECAM1 labels germ cells and endothelial cells; AMH labels Sertoli cells within testis cords.



