Programming Eosinophils in the Esophagus
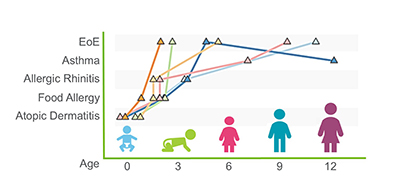
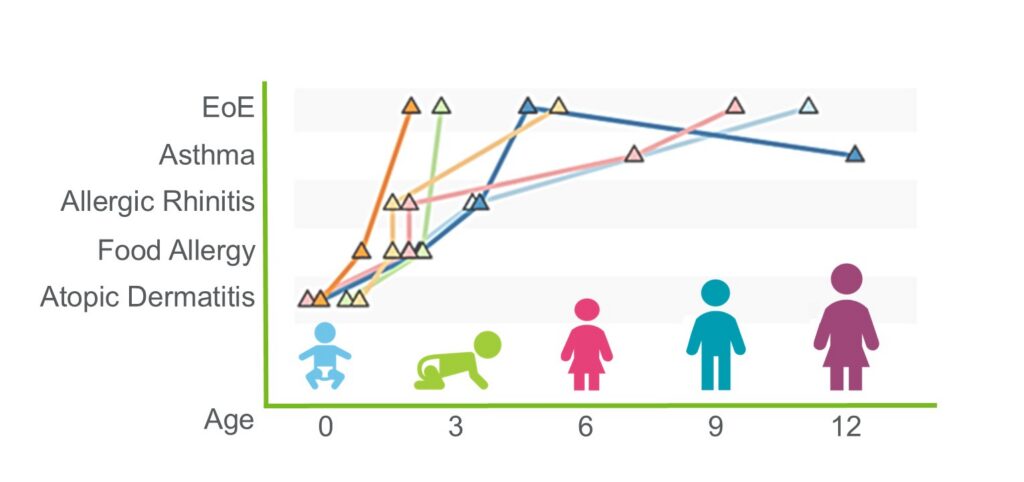
Research from the Rothenberg CURED Lab published in Nature Communications demonstrates that eosinophils residing in the bone marrow and the esophagus are readily distinguishable by the marked organization of their chromatin landscape, gene expression, and surface immunophenotype. Esophageal eosinophils are programmed by thousands of different molecular events, demonstrating the complexity of eosinophilic inflammation.
“The acquisition of tissue specialization at the molecular level is remarkably conserved across species according to mouse and human single-cell RNA sequencing findings. Mouse esophageal eosinophils have a distinct tissue-specific epigenome that is enriched for genes whose human counterparts are associated with allergic disease risk loci and eosinophilia. We have molecular evidence that esophageal eosinophils exist as at least two populations and that eosinophil tissue phenotypes are a result of local environmental cues,” says Jennifer Felton, PhD, the first author.

“Interestingly, the transcription factors involved in tissue specialization are largely distinct from those known to be involved in eosinophil development. We found that a complex of transcription factors, including AP-1 family members, are involved in acquiring the esophageal phenotype of eosinophils. Our reported findings and the genomic datasets now released to the public are facilitating a better understanding of eosinophils in the context of allergic and esophageal inflammation,” says Marc Rothenberg, MD, PhD, the senior author.